[Physics FAQ] -
[Copyright]
By Don Koks, 2003.
What are Half Lives and Mean Lives?
Specifying the half life or mean life of a process is a way of quantifying how fast it
is occurring, when the whole process would in principle take forever to complete.
The example we will talk about here is radioactive growth and decay, but examples from
other fields include the recovery of a muscle after some exertion, and the filling of a
cistern.
In particular then, the half life of a radioactive element is the time required for
half of it to decay (i.e. change into another element, called the "daughter" element).
So if a radioactive element has a half life of one hour, this means that half of it
will decay in one hour. After another hour, half of the remaining material will
decay. But why didn't all of that remaining material decay in that second
hour? Does the element somehow know that it's decaying, and alter its decay speed to
suit?
Textbooks are usually content with deriving of the law of decay, and don't tend to
address this question. And yet it forms a classic example of the way in which
research in physics (and science in general) is carried out. Regardless of how we
might expect an element to behave—where perhaps the second half might be
expected to decay in the same amount of time as the first half—this simply does not
happen. We must search for a theory that predicts this.
Science is often thought to proceed by our logically deducing the laws that govern the
world. But it's not that simple; there are limits to what we can deduce, especially
about things in which we cannot directly participate. Radioactive decay is a good
example of this. We can't use a microscope to watch the events that make an element
decay. The process is quite mysterious. But what we can do is make a simple
theory of how decay might work, and then use that theory to make a prediction of what
measurements we can expect. That's the way science proceeds: by making theories that
lead to predictions. Sometimes these predictions turn out to be wrong. That's
fine: it means we must tinker with the theory, perhaps discard it outright, or maybe
realise that it's completely okay under certain limited circumstances. The hallmark
of a good scientific theory is not what it seems to explain, but rather what it
predicts. After all, a theory that says the universe just appeared
yesterday, complete with life on Earth, fossils and so on, in a sense "explains"
everything beautifully by simply defining it to be so; but it predicts absolutely
nothing. So from a scientific point of view it is not a very useful theory, because
it contains nothing that allows its truth to be tested. On the other hand, while
it's arguable that the theory of quantum mechanics explains anything at all, it certainly
does predict a huge number of different phenomena that have been observed; and
that's what makes it a very useful theory.
For radioactive decay, our theory is that the atoms decay, or change into another atom,
quite spontaneously. At a basic level, we don't know why this should be; but we can
only proceed step by step, and so first we begin with this simple theory. We
postulate that they decay independently of whether their neighbours are decaying, and also
that their tendency to decay is independent of how old they are. A given atom might
decay after one microsecond, or one million years. However long it has been sitting
intact makes no difference to its ability to decay right now. If the mechanism
behind its decay is strong, in the sense that the atom has a large chance of decaying,
then it won't last long: after all, the chance that it won't decay in some time
interval is small, so the chance that it survives for any appreciable amount of time is
then also small. That can be worked out by simple probability: multiplying together
the probabilities that it doesn't decay for a string of those time intervals. The
statistics of decaying elements, such as the mean and standard deviation of the number of
atoms decaying in various time intervals, were measured soon after radioactivity was
discovered; they were found to match those predicted by this idea of random decay, called
Poisson statistics. Whenever anything has a small chance of happening, but
there are lots of opportunities for it to happen, we get Poisson statistics.
So certainly physics has not proven, and can never prove, that its theory of
atomic decay is true. The logical process is that if atoms decay randomly,
then Poisson statistics will result. Experiments show that Poisson
statistics do indeed result, but logically this does not mean that atoms decay
randomly. Nevertheless, the way of science is that we do postulate that atoms decay
randomly, until a new experiment calls this into question. But no experiment
ever has. If this sounds like a reverse use of logic, then consider the same ideas
for mechanics. Ideas of gravity, mass and acceleration were originally produced by
Newton through the same process: because they predicted planetary orbital periods that
could be verified experimentally. Because of this great success, expressions such as
F = ma and F = GMm/r2 came to be canonical in physics.
The logic was indeed being used in reverse; but no one was surprised when, three centuries
later, one of the moon astronauts dropped a feather and a hammer together in the moon's
vacuum, and found that they both fell at the same rate (although it was still beautiful
and dramatic to watch!). That reverse logic had, after all, allowed him to get to
the moon in the first place. So this way of conducting science works very well.
A "people" experiment to simulate radioactive decay
We can perform a simple test of this theory of radioactive decay by doing an experiment
with a big group of people; the group should be large so as to get good statistics.
Put 1000 people into a big hall, and give each a coin. Each person represents a
radioactive atom, and the coin represents their ability to decay. Tell everybody to
toss their coin once per minute. If the result is heads, then the person should
immediately leave the hall (which corresponds to the atom decaying). If tails, then
take no action, except waiting for another minute to elapse, and then tossing again.
What happens? After one minute, roughly half of the people get up and walk out,
because their coins landed heads up. After another minute, everyone tosses again,
and about half the remainder will walk out. Of course we don't expect
everyone to leave after the second minute; as each minute goes by, roughly half
of the group walks out. Our simple model of random behaviour has produced a half
life! We say that this particular "element" has a half life of one minute.
Really of course, the laws of physics don't operate at one minute intervals; we should
really ask everyone to toss continuously. This is fine, but it would cause a lot of
commotion and make it harder to see what was happening.
Although it's not obvious, it can be shown that in such a situation where the amount of
an element halves in a constant time interval, it will also "third" in a different
constant time interval that is not hard to calculate. In fact, we can choose any
number we like to describe the decay speed. Choosing the halving time is
conventional because it's unambiguous. If a radioactive element was said to have a
"third life" of one hour, then no one would ever remember if that means that after one
hour one third has decayed, or one third is left over. Specifying
the half life doesn't produce this problem of course.
Getting back to our group of 1000 people, still flipping their coin every minute, we
could change its half life by changing the requirement to leave. For example, every
minute each person tosses their coin quickly twice. But now, only those getting two
heads must leave. In that case, after the first minute, one quarter will leave, and
after the second minute, one quarter of the remainder will leave. The half life now
is longer than one minute. So, we've arranged for this "element" to decay more
slowly, by lowering the chance that any one of its constituent atoms decays.
Remember that any particular person who sits and tosses their coin might sit for any
amount of time, irrespective of how many of their neighbours have now left the hall.
They might even sit for years continually flipping their coin, only to find it always
lands tails up. The chance is small of course that this will happen, but it just
might. The chance that any person leaves is completely independent of how long they
have been sitting there.
A "mustard seed" experiment to simulate radioactive decay
Here is another way of demonstrating radioactive decay, this time closer to the feel of
a real experiment. Put a couple of teaspoons of mustard seeds in a mortar, and grind
them with a pestle. For a classroom demonstration, have a microphone nearby that
amplifies the click as each seed bursts. In the random crashing-together of the
seeds, the chance that any particular one will burst is roughly independent of its
neighbours and also roughly independent of time. So what we hear is initially a
burst of clicks that soon dies down, with a half life of several seconds or longer.
This is just like what would be heard if we put a Geiger counter next to a rapidly
decaying element.
The mean life of a process
Given our radioactive element, if half of its atoms have decayed after one half life,
then we can expect there to be some kind of well defined average life expectancy: the
mean life of the atoms, which is somewhat longer than their half life. It
turns out that the mean life equals the half life divided by the natural logarithm of 2
(about 0.693). The mean life also turns out to exactly equal the number τ
that appears in the exponential term e−t/τ involved with describing
decay or growth, called the time constant. The label "time constant" is
just a name for τ; the fact that the mean life happens to equal τ is
a kind of neat coincidence that allows us to think of the mean life in the following,
alternative, way.
We said above that the speed of any decay process can be described by the time it takes
for the remaining amount of the element to fall to a half, or a third, or any fraction at
all. One special choice of that number is e, and the length of time after
which the amount remaining has been reduced to 1/e of the original also happens
to equal the mean life of the decay. This is one reason why the number e is
so special. Besides the fact that e has friendly properties that simplify
the mathematics of growth and decay, it also quantifies such an everyday idea as the
average life expectancy of the atoms.
When we plot the amount of a radioactive element as a function of time, we find that it
drops with a characteristic "exponential decay curve" that helps to show this half life
idea more mathematically. Here is a question: 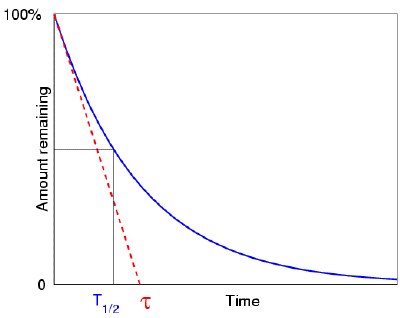 suppose that the element did not decay
exponentially (i.e. with a half life), but instead decayed linearly, meaning its rate of
decay was always equal to its initial rate (giving rise to a simple "first half in one
minute and the second half in the next minute—and then, all gone" sort of idea). In
such an idealised case, how long would it take to completely vanish? That is,
suppose we have 1000 atoms of a radioactive element, and initially there are 10 atoms
decaying per second. If it was to keep decaying at this rate (it won't, but
imagine that it did), then how long would it take to completely vanish? The answer
is of course 100 seconds. Now it turns out that this is precisely equal to the mean
life of the real element. (Which means that we know straight away that the real
element's half life is 69.3 seconds.)
suppose that the element did not decay
exponentially (i.e. with a half life), but instead decayed linearly, meaning its rate of
decay was always equal to its initial rate (giving rise to a simple "first half in one
minute and the second half in the next minute—and then, all gone" sort of idea). In
such an idealised case, how long would it take to completely vanish? That is,
suppose we have 1000 atoms of a radioactive element, and initially there are 10 atoms
decaying per second. If it was to keep decaying at this rate (it won't, but
imagine that it did), then how long would it take to completely vanish? The answer
is of course 100 seconds. Now it turns out that this is precisely equal to the mean
life of the real element. (Which means that we know straight away that the real
element's half life is 69.3 seconds.)
This way of looking at the concept of mean life also appeals to our intuition: it says
that if the atoms behaved in the nice, linear way that we humans love to think
in, then the time needed for them all to decay would be exactly equal to the mean life of
the actual, real world, element. You can see that the number e is tied in
to simple ideas of linearity, and that's another reason why it is such a powerful number
in mathematical analysis.
This same idea applies to the growth of the daughter element. Suppose for
simplicity that the daughter is not itself radioactive. Initially there are no
daughter atoms, and gradually they grow as the parent element decays. Their growth
must eventually flatten out, and this takes infinite time, for the same reason that it
takes infinite time for the parent element to fully decay. We ask the question:
if the growth of the daughter continued at its initial rate, (which it doesn't,
remember!), then how long would it take for the sample to be completely composed of
daughter atoms? Again, this time interval turns out to be the mean life of the
parent.
The mean life and "probability per second"
That last way of applying the mean life to growth produces a way of speaking that is
not always well understood by those who use it. Suppose we have a single atom of a
radioactive element, sitting in front of us, waiting to decay. We ask: what is the
probability that after some time t, it has decayed? On the average, we can
expect that after one mean life τ has elapsed, there's a reasonable chance that
the atom will have decayed. Although it might take a million times that period
before it actually does decay, we feel confident that after perhaps two or three mean
lives have passed, the atom will almost certainly have decayed.
We cannot really plot this probability as a function of time, because that would
require knowing when the atom will decay (since then the probability equals one).
So, the best we can do is to make a generic plot that correctly describes the whole
population of decaying atoms, since the atom in front of us behaves just like the other
atoms of the element. In that case, we expect that the probability will grow,
tending towards one as t goes to infinity. Remember: this is an average
sort of prediction; any individual atom will certainly attain one at some point. But
it's the best we can do.
The actual expression for this probability turns out to be 1 −
e−t/τ, which indeed tends toward one as t tends to
infinity. (It's actually the same curve as that describing the growth of the
daughter element.) 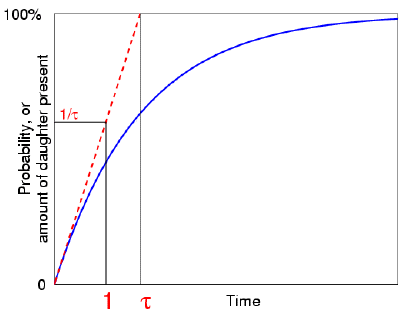 Now, what is the value of this (generic) probability when
t=1: what is the chance that the atom has decayed after one unit of time?
Well, the answer is 1 − e−1/τ. . . which might not
communicate very much! So as we did before, let's ask another, related,
question. If the probability of decaying within time t continued
to increase at its initial rate, (which it doesn't really!), then how long would it take
for the atom to decay—after how long would this probability reach one? Again,
this time interval turns out to be the mean life τ. But if our linear,
simplified, atom reaches decay probability one after a time τ, then it must
reach probability 1/τ after a unit time.
Now, what is the value of this (generic) probability when
t=1: what is the chance that the atom has decayed after one unit of time?
Well, the answer is 1 − e−1/τ. . . which might not
communicate very much! So as we did before, let's ask another, related,
question. If the probability of decaying within time t continued
to increase at its initial rate, (which it doesn't really!), then how long would it take
for the atom to decay—after how long would this probability reach one? Again,
this time interval turns out to be the mean life τ. But if our linear,
simplified, atom reaches decay probability one after a time τ, then it must
reach probability 1/τ after a unit time.
In our example above with 1000 atoms and initially 10 decays per second, we concluded
the mean life was τ = 100 seconds. So, if the chance that any
particular atom had decayed was to keep increasing uniformly at its initial rate, then
after one second, the chance that it had decayed would be 1/100. This makes sense:
1/100 of the initial 1000 atoms is the 10 atoms we measured to have decayed. So we
say "the atom has a probability of decay of 1/100 per second". Remember, this
doesn't mean that after 100 seconds it will definitely have decayed! This would only
be true if the atom were to behave in a simple, linear, way.
The fact that the atom's probability of having decayed is "slowing down", is just like
a pushed trolley that starts out with a speed of 1/100 metre per second, but decelerating
due to friction at just the right rate so as to match our atom. If there were no
friction so that it moved at constant speed, then it would take 100 seconds to travel a
metre. It doesn't do this of course—it never quite reaches a metre distance because
it continuously slows down; but 1/100 m/s refers to its initial rate of distance
increase. And likewise for our generic atom that represents the whole population of
atoms, "1/100 per second" refers to its initial rate of "decay-probability
increase". In an average sense for the whole population, that decay probability will
never quite reach one, although it will eventually reach one for any particular
atom.
This whole discussion of growth can also be applied to the daughter element, because
that grows with precisely the same time constant τ. If it continued to
grow at its initial rate, then after a time τ the sample would be completely
composed of daughter atoms.
You can see that we can juggle these numbers and get a feel for the details of an
element's decay or growth. But for these more refined ideas, it's certainly a whole
lot easier to think in terms of the mean life, than the half life. Even so, both the
mean life and the half life give us an intuitive feel for radioactive decay, and each has
its own domain of usefulness.
 suppose that the element did not decay
exponentially (i.e. with a half life), but instead decayed linearly, meaning its rate of
decay was always equal to its initial rate (giving rise to a simple "first half in one
minute and the second half in the next minute—and then, all gone" sort of idea). In
such an idealised case, how long would it take to completely vanish? That is,
suppose we have 1000 atoms of a radioactive element, and initially there are 10 atoms
decaying per second. If it was to keep decaying at this rate (it won't, but
imagine that it did), then how long would it take to completely vanish? The answer
is of course 100 seconds. Now it turns out that this is precisely equal to the mean
life of the real element. (Which means that we know straight away that the real
element's half life is 69.3 seconds.)
suppose that the element did not decay
exponentially (i.e. with a half life), but instead decayed linearly, meaning its rate of
decay was always equal to its initial rate (giving rise to a simple "first half in one
minute and the second half in the next minute—and then, all gone" sort of idea). In
such an idealised case, how long would it take to completely vanish? That is,
suppose we have 1000 atoms of a radioactive element, and initially there are 10 atoms
decaying per second. If it was to keep decaying at this rate (it won't, but
imagine that it did), then how long would it take to completely vanish? The answer
is of course 100 seconds. Now it turns out that this is precisely equal to the mean
life of the real element. (Which means that we know straight away that the real
element's half life is 69.3 seconds.) Now, what is the value of this (generic) probability when
t=1: what is the chance that the atom has decayed after one unit of time?
Well, the answer is 1 − e−1/τ. . . which might not
communicate very much! So as we did before, let's ask another, related,
question. If the probability of decaying within time t continued
to increase at its initial rate, (which it doesn't really!), then how long would it take
for the atom to decay—after how long would this probability reach one? Again,
this time interval turns out to be the mean life τ. But if our linear,
simplified, atom reaches decay probability one after a time τ, then it must
reach probability 1/τ after a unit time.
Now, what is the value of this (generic) probability when
t=1: what is the chance that the atom has decayed after one unit of time?
Well, the answer is 1 − e−1/τ. . . which might not
communicate very much! So as we did before, let's ask another, related,
question. If the probability of decaying within time t continued
to increase at its initial rate, (which it doesn't really!), then how long would it take
for the atom to decay—after how long would this probability reach one? Again,
this time interval turns out to be the mean life τ. But if our linear,
simplified, atom reaches decay probability one after a time τ, then it must
reach probability 1/τ after a unit time.