Where do the normal and anomalous Zeeman formulas come from?
First, a bit of classical physics. Imagine an ``infinitesimally small''
bar magnet - this is called a magnetic dipole. A vector (say
![]() ) represents the strength and direction of the dipole;
) represents the strength and direction of the dipole;
![]() is known as the magnetic moment.
is known as the magnetic moment.
Suppose we place the dipole in a magnetic field B. The field will
exert a torque on the dipole, trying to flip it into alignment (so B
and ![]() are parallel). Twisting the dipole out of alignment
takes energy. In other words, the dipole has a potential energy that
depends on its orientation with respect to the field. Classical physics
says that this ``magnetic'' energy is given by
are parallel). Twisting the dipole out of alignment
takes energy. In other words, the dipole has a potential energy that
depends on its orientation with respect to the field. Classical physics
says that this ``magnetic'' energy is given by
![]() .
.
A tiny current loop acts like a dipole, with a magnetic moment
perpendicular to the plane of the loop. The current loop possesses angular
momentum (after all, the current consists of moving charges, which have
mass). According to classical physics, the magnetic moment is proportional
to the angular momentum:
![]() constant times J. The
constant depends on the charge/mass ratio of the moving charges making up
the current.
constant times J. The
constant depends on the charge/mass ratio of the moving charges making up
the current.
We now apply all this to an electron in an atom in a magnetic field.
Thanks to its orbital angular momentum l, it has a ``magnetic''
energy proportional to
![]() ; likewise, the spin
contributes energy proportional to
; likewise, the spin
contributes energy proportional to
![]() . Adding up the
l's and s's for all the electrons in the atom, we might expect
a formula like this for the total ``magnetic'' energy:
. Adding up the
l's and s's for all the electrons in the atom, we might expect
a formula like this for the total ``magnetic'' energy:
Without the enigmatic 2, we'd simply have ![]() on the right hand side of
formula 7. Without
on the right hand side of
formula 7. Without ![]() on the right hand side, the normal and
anomalous Zeeman formulas would be the same-- as we will see.
on the right hand side, the normal and
anomalous Zeeman formulas would be the same-- as we will see.
How does the electron maintain its orientation in the magnetic field-- why doesn't it snap into alignment? Or in ``old quantum'' terms, how can we have stationary states with S (or L) out of alignment with the field? This was a legitimate question for the old quantum theory, and it had a good answer. The spinning electron acts like a gyroscope. The field exerts a torque on the electron, and like any gyroscope, the electron precesses. (The same argument holds for L.)
We can get the ``anomalous Zeeman'' formula from formula 7 with a little vector algebra, and a crucial assumption:
Assume that in any stationary quantum state, S and L each precess around J, so that the projection of S on J doesn't change, but the component of S perpendicular to J rotates uniformly (ditto for L).This is called spin-orbit coupling. Only without an external magnetic field is it strictly true. It is approximately true if the external field is weak enough. It breaks down in a strong magnetic field, a phenomenon known as spin-orbit decoupling.
Where does spin-orbit coupling come from? Naively expressed, from a torque
trying to make L and S point opposite to each other (i.e.,
anti-parallel). Goudsmit and Uhlenbeck imagined it this way. Suppose
there is only a single electron. If we ride along an electron's orbit
right next to it, but not spinning ourselves, we will appear to see the
nucleus orbiting us (shades of Ptolemy!) Effectively we have a
positive current circulating around us; this will generate a magnetic field
proportional to L. The electron has a magnetic moment proportional
to S, and hence we get an energy term proportional to
![]() . (Because of the positive charge on the nucleus, the signs
come out so that spin-orbit coupling trys to make L and S
anti-parallel.)
. (Because of the positive charge on the nucleus, the signs
come out so that spin-orbit coupling trys to make L and S
anti-parallel.)
Life is more complicated in multi-electron atoms.
![]() is
only an approximation, applicable when the electron-electron forces
dominate the spin-orbit forces. In this case, one can approximate the
totality of electrons as a single electron ``smear'', with total orbital
angular momentum L, and total spin S. For ``light'' atoms,
this works pretty well. For ``heavy'' atoms, a different approximation
works better.
is
only an approximation, applicable when the electron-electron forces
dominate the spin-orbit forces. In this case, one can approximate the
totality of electrons as a single electron ``smear'', with total orbital
angular momentum L, and total spin S. For ``light'' atoms,
this works pretty well. For ``heavy'' atoms, a different approximation
works better.
A classical model has emerged. The vectors L and S feel a
torque trying to make them anti-parallel; when the magnetic field B
is turned on, L and S each feel a torque trying to align them
with B. We have, in other words, ``perturbation terms'' occuring
somewhere in the total energy formula:
Classical mechanics would next try to compute the ``trajectories'' of the
vectors L and S (i.e., how they vary with time). If ![]() , then J is constant (conservation of angular momentum), and
L and S precess about J. If the field is weak, then (to
a good approximation) J precesses about B, while L and
S still precess about J. J is no longer constant, since
it is subject to an external torque. If the field is strong, then L
and S precess about B (again to a good approximation), because
the magnetic torque swamps the spin-orbit torque.
, then J is constant (conservation of angular momentum), and
L and S precess about J. If the field is weak, then (to
a good approximation) J precesses about B, while L and
S still precess about J. J is no longer constant, since
it is subject to an external torque. If the field is strong, then L
and S precess about B (again to a good approximation), because
the magnetic torque swamps the spin-orbit torque.
The ``old quantum'' prescription says to find the energy levels of these stationary states.
As it happens, the energy of a stationary state depends primarily on terms
other than those in formula 8. We have terms analogous to the
![]() of hydrogen; then terms depending on the magnitude of L and
S (i.e., terms depending on
of hydrogen; then terms depending on the magnitude of L and
S (i.e., terms depending on ![]() and
and ![]() ); only after that does
formula 8 come into play.
); only after that does
formula 8 come into play.
The spin-orbit coupling contributes a perturbation that depends on the
angle between L and S (and hence indirectly on the magnitude of
J). So when ![]() , we expect to find bunches of energy levels
with the same
, we expect to find bunches of energy levels
with the same ![]() and
and ![]() grouped closely together, separated slightly by
an energy difference depending on
grouped closely together, separated slightly by
an energy difference depending on ![]() . This is an aspect of the famous
fine structure of the spectrum.12 If
. This is an aspect of the famous
fine structure of the spectrum.12 If
![]() , then the fine structure disappears. (Low-lying energy levels of
atoms with even numbers of electrons usually have
, then the fine structure disappears. (Low-lying energy levels of
atoms with even numbers of electrons usually have ![]() .)
.)
Without a field, we have complete rotational symmetry in the energy
formula. The spin-orbit perturbation depends only on the magnitude
of J, not on its direction (on ![]() , not on
, not on ![]() ). With a field, the
symmetry is broken and the degeneracy lifted.
). With a field, the
symmetry is broken and the degeneracy lifted.
We have perturbations depending on
![]() and
and
![]() . With a strong field (or if
. With a strong field (or if ![]() ), we confidently replace
these dot products with
), we confidently replace
these dot products with ![]() and
and ![]() , for the dot products don't
change and must be quantized (i.e., they are stationary). We have seen
already how this leads to the normal Zeeman effect. Note also another
regularity: spectra without fine-structure have
, for the dot products don't
change and must be quantized (i.e., they are stationary). We have seen
already how this leads to the normal Zeeman effect. Note also another
regularity: spectra without fine-structure have ![]() , and hence display
normal Zeeman splitting.
, and hence display
normal Zeeman splitting.
With a weak field, we express the perturbation in terms of
![]() and
and
![]() . The first term we replace with
. The first term we replace with
![]() . Were this all, we would again see a normal Zeeman effect, because
. Were this all, we would again see a normal Zeeman effect, because
![]() and
and ![]() satisfy the same selection rule.
satisfy the same selection rule.
Turn to the
![]() perturbation. Because S precesses
about J, the average value of
perturbation. Because S precesses
about J, the average value of
![]() is the same
as
is the same
as
The rest is vector algebra. You may skip to the next section if you like-- no new concepts appear.
First derive a formula for
![]() , in terms of
quantum numbers. We have:
, in terms of
quantum numbers. We have:
Next derive a formula for
![]() , the projection of S on
J:
, the projection of S on
J:
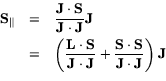
© 2001 Michael Weiss